 
While the resistance of the mutants to NaCl was unaltered, their detergent resistance was decreased under both growth and non-growth conditions. 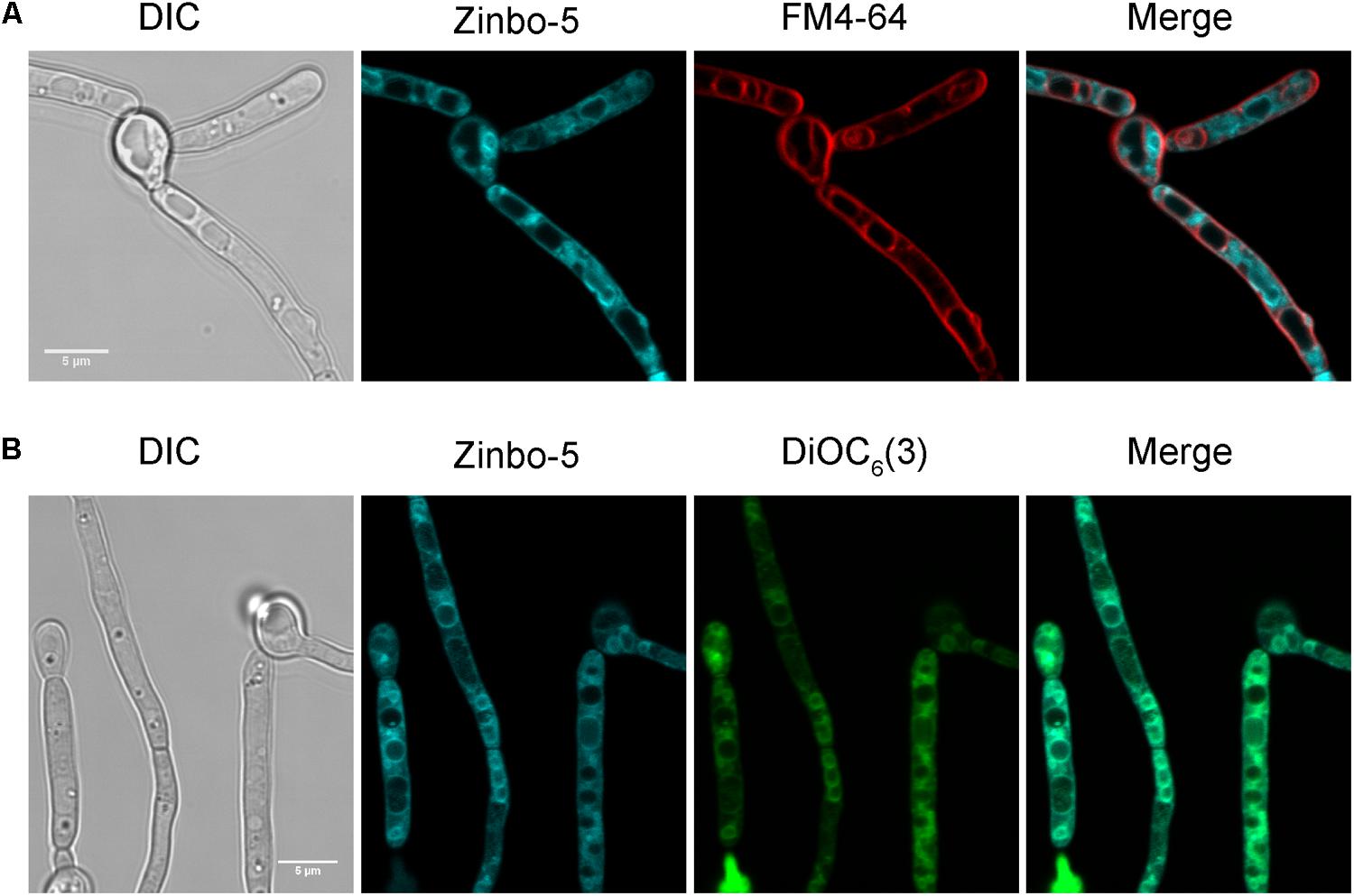
The mutant populations synthesized much less cyclopropane fatty acids and were more resistant than the wild type to freezing and thawing in saline only, exhibiting less viability loss and less wall and membrane damage. Strains of Escherichia coli proficient and deficient in cylopropane fatty acid synthesis were compared for fatty acid content, cryosensitivity, presence of freeze-thaw-induced wall and membrane damage, resistance to detergent-stimulated lysis and tolerance to salt and detergents during growth. Journal of Applied Bacteriology56, 165–172. Cryosensitivity of Escherichia coli and the involvement of cyclopropane fatty acids. These results indicate that the cAMP-CRP complex exerts negative transcriptional control of ODC synthesis as a function of the speC promoter.Ĭalcott, P.H. However, when a isogenic crp- strain was supplemented with 2 mM cAMP, no repression of ODC mRNA was observed. When a cya- strain was supplemented with 2 mM cAMP, steady state levels of ODC mRNA were repressed by 80%. In contrast, in crp- strains transformed with pCOD or pTET (TcR), cAMP supplementation did not change their Tc resistance. In a prototroph transformed with the plasmid pCOD bearing a speC::tet chimeric gene, cAMP supplementation decreased tetracycline (Tc) resistance. This repression required a functional CRP as cAMP failed to repress ODC synthesis in vitro in an extract prepared from a crp- strain the addition of purified CRP to the crp- extract restored the ability of cAMP to repress ODC synthesis. In a cell-free protein synthesizing system directed by pODC, cAMP at 10(-5) M repressed ODC synthesis by 90%. 
In minicells transformed with the plasmid pODC bearing speC, cAMP supplementation repressed ODC synthesis. The speC gene encoding ornithine decarboxylase (ODC) in Escherichia coli is negatively regulated by cAMP and the cAMP receptor protein (CRP). We suggest that expression of other recombinant genes directed by various promoters that allow permanent transcription during growth with limited glucose supply in chemostat and fed-batch fermentors can be improved by appropriate insertion of the cap region. 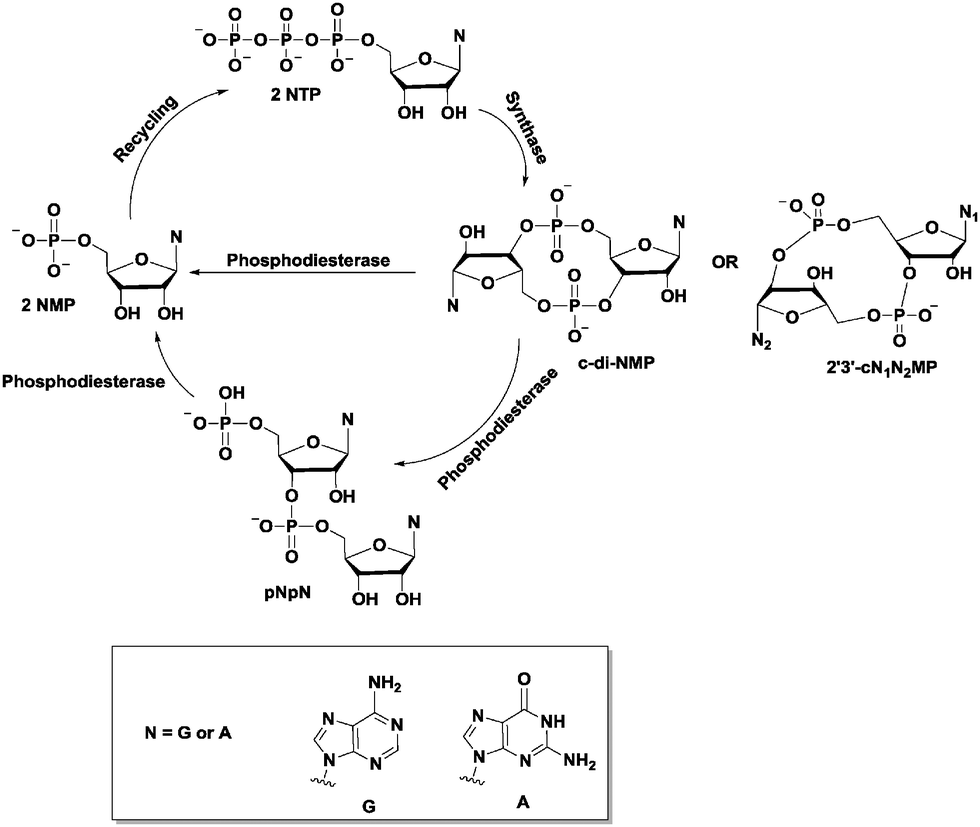
Determinations of plasmid copy numbers, cellular amounts of galactokinase-mRNA, activity of galactokinase (AGalK) and the concentration of galactokinase at various dilution rates (D) strengthen the conclusion that the increase in AGalK with decreasing D was indeed due to the cap-mediated enhancement of transcription of the galK gene. coli TG1(pDR720) could be improved in the same way with cap-trpP.O-galK in E. In addition, expression of the galactokinase gene (trpP.O-galK) in E. These results are in line with the increase in the level of cAMP with declining dilution rate and the well-known positive effects of cAMP-catabolite gene activator protein (CAP) at the transcriptional level. The cap-mediated stimulatory effect on the expression of cap-trpP.O-ifn alpha 1 increased with decreasing dilution rate. coli TG1(pHRW500cap) in glucose-controlled continuous and fed-batch cultures. The expression of ifn alpha 1 could be improved after insertion of the catabolite activator region (cap) upstream to trpP.O during cultivation of the modified E. Often the only data that support cAMP involvement in these processes includes cAMP measurement, detection of the enzymes involved in cAMP metabolism, or observed effects of high concentrations of the nucleotide on cell growth.Įscherichia coli TG1 (pHRW500) permanently expressed the human interferon alpha 1 gene (ifn alpha 1) directed by the tryptophan promoter (trpP.O) during continuous and fed-batch cultivation with a limited supply of glucose. A role for cAMP has been suggested in nitrogen fixation. cAMP has been implicated in regulating antibiotic production, phototrophic growth, and pathogenesis. For other bacteria, less detail is known. This review summarizes recent work focused on elucidating the molecular mechanisms of CRP-cAMP complex-mediated processes. Enteric coliforms have provided a model to explore the mechanisms involved in controlling adenylate cyclase activity, in regulating adenylate cyclase synthesis, and in performing detailed examinations of CRP-cAMP complex-regulated gene expression. The CRP-cAMP complex subsequently binds specific DNA sequences and either activates or inhibits transcription depending upon the positioning of the complex relative to the promoter. In the enteric coliforms, the effects of cAMP on gene expression are mediated through its interaction with and allosteric modification of a cAMP-binding protein (CRP). cAMP plays a role in regulating gene expression, not only for the classic inducible catabolic operons, but also for other categories. Cyclic AMP (cAMP) is found in a variety of prokaryotes including both eubacteria and archaebacteria. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
